One critical component of turning a vision of a life-saving device into reality is found in the development of a working prototype. One that moves beyond the conceptual to take the science behind the device—in this case, a technology that detects fetal arrhythmia in pregnant mothers—and applies human factors design and mechanical engineering solutions to it as a proof of viability.
Early detection of fetal arrhythmia can identify anomalies and potential life-threatening complications during fetal development, helping to save lives. After securing a grant to develop a fetal magnetocardiography (fMCG) solution—essentially sensors that record the magnetic fields generated by the electrical activity of the heart and thus assess the health of the fetal heart while still in the womb—client, Applied Physics Systems (APS), turned to Mighty Studios for the next phase.
Their ask? Take the idea and iron out the practical and human factors considerations of not just how it looks and functions, but how it might operate within the day-to-day setting of a hospital.
The challenges Mighty faced ranged from addressing the physical, ergonomic, and human interaction aspects of the machine (how a pregnant mother might get onto the patient bed/table, for example), to the mechanical engineering practicalities of sensor placement and how to cater to the size variables found between early and late term pregnancies.
The basics of how electrical components and wiring were positioned and installed, plus how the patient conveyance system (the table) slid into the Cylindrical Shield System (the chamber) for readings to occur, were also solved during development.
Over the course of an 18-month Research and Development phase, Mighty drew on an extensive background in human factor design to develop multiple iterative prototypes addressing these challenges and more, setting up Applied Physics Systems to take their next steps in introducing this technology into hospitals as part of the treatment and monitoring timeline of modern pregnancy.
INSIGHTS
“Fetal arrhythmia is a significant cause of fetal demise and unexplained fetal deaths.
While most occurrences are benign, serious arrhythmia can lead to life-threatening complications if undiagnosed or untreated.”
These words, taken from the project outline provided by client, APS, set the scene for what’s at stake with this project. By detecting early, the need for surgery on a newborn baby with fetal arrythmia can be greatly reduced and many lives saved.
With the technology of it—an integrated OPM-based fMCG/ferromagnetic shield system—proven feasible, Mighty Studios set out to tackle the physical mechanics of the device, focusing on understanding the ergonomic inputs and constraints to visualize a solution. For Mighty, it’s more than just what something looks like; it’s taking into consideration how people (both patient and operator) will interact with it.
Top of mind was the simple statement: Pregnant mothers need to be handled with care.
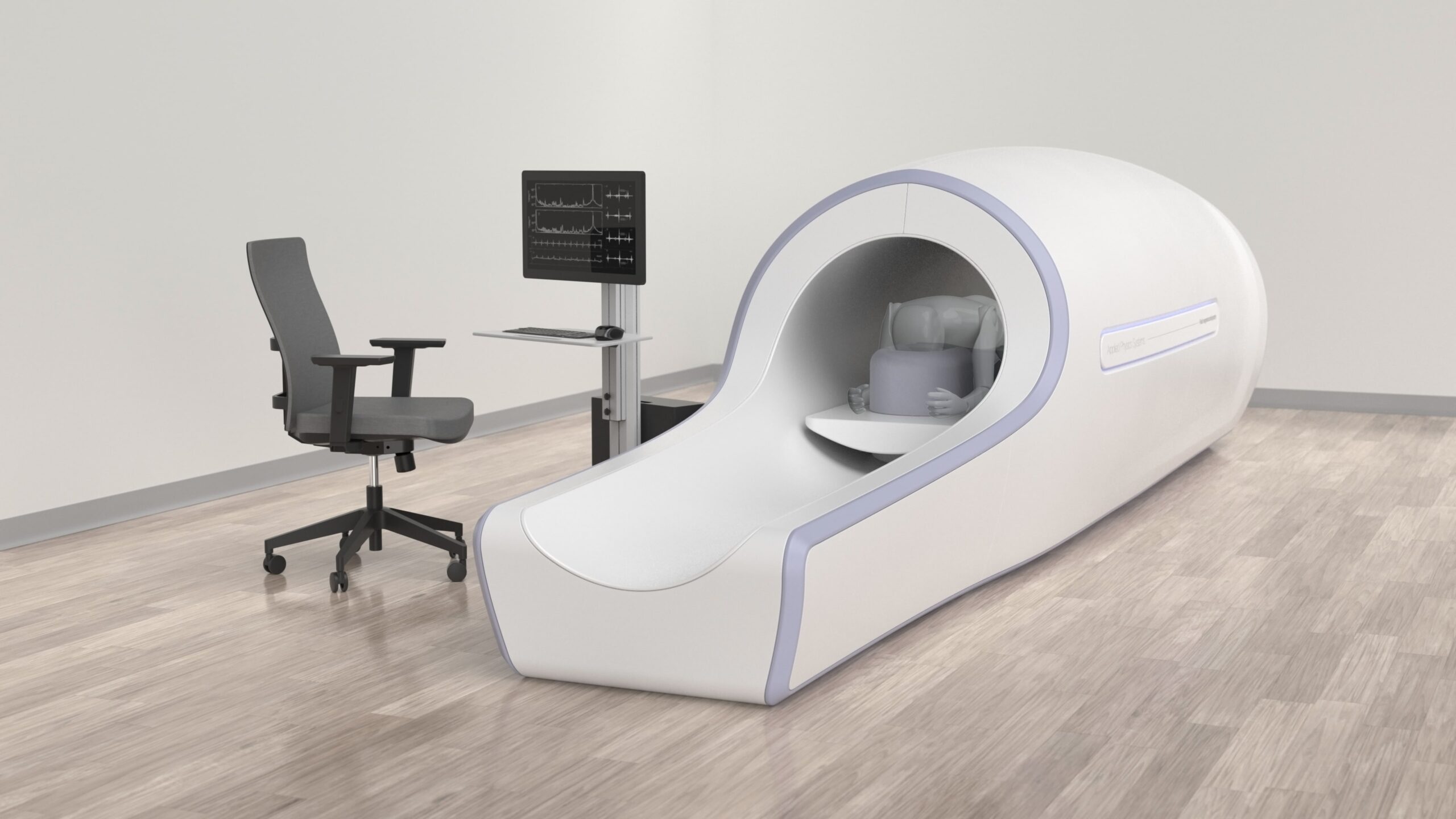
Perhaps the biggest challenge is that they’re not the most dexterous, especially late-term mothers. Depending on where they are in their pregnancy, their size and maneuverability are affected greatly. In working on the patient conveyance system (the bed that would slide into the chamber), Mighty knew that their solution had to cater to women at all stages of the development cycle and provide a safe and comfortable experience at that first step: getting on and off the table.
While APS had developed a basic prototype early on, the first adjustment Mighty made was a simple one: changing the height of the patient bed. Low access means easy access. Knowing that the standard height of a wheelchair is 17 inches off the ground, Mighty’s solution allowed for a heavily pregnant mother to be wheeled into the room and simply roll onto the table from that wheelchair.
The next consideration was the adjustable cradle that the belly would be held in while the readings were taken. This apparatus allowed the patient’s stomach to be positioned above the sensor array for accurate readings and, critically, provided a comfortable and safe experience for the expectant mother.
Learning as they prototyped, Mighty utilized a dummy human form to test positioning, but also took it a step further and purchased a pregnancy suit for in-house testing. This enabled them to conduct rudimentary testing while developing the cradle. The team also consulted frequently with physicians, many of them female, to provide insight and advice on testing to account for all variables during pregnancy.
The table/cradle combination was based on a massage table concept whereby the patient lies face down on a padded surface, but in this case, with the supported stomach hanging down through an opening designed to suspend and position the fetus over the sensor array. This support cradle solution—an adaptable arch design—is fully adjustable depending on the size of the expectant mother, opening wider or narrower like the aperture in a camera lens.
From an engineering perspective, development of this cradle required Mighty to consider the mechanics of where the technology was housed; how to keep the mother comfortable, supported, and calm with no pinch points or hard edges on the cradle; and how to ensure the fetus was positioned as close to the sensors as possible. Comfort was addressed through a combination of thick foam padding and neoprene, along with hand/arm positioning options and, like a massage table, support for the face.
With the stomach hanging down, the fetus could drop the lowest point, as close to the sensors as possible, gently touching the surface of the belly. With so many variables in play—the baby can be higher or lower depending on the woman and pregnancy progression—adjustability was also a critical consideration.
While the sensors—individual magnetometers roughly the size of a thumb—were positioned in an array to take the reading, Mighty was challenged with how to connect the array and wires throughout the system. By creating a safe path for the cables within the device, trip hazards were reduced and safety risks mitigated.
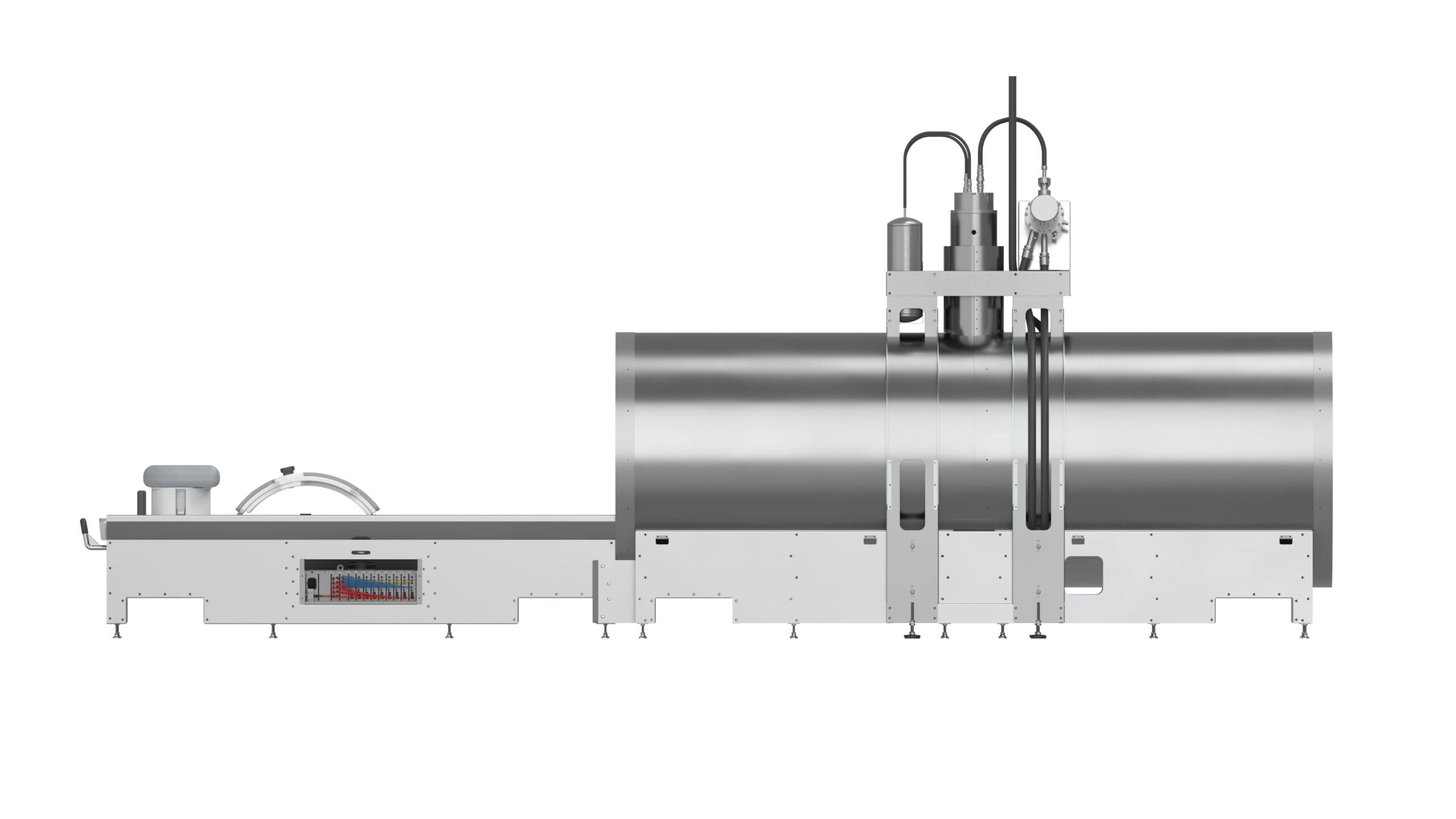
Once the human factors were addressed, the next item on the list was the mechanics of how the bed would slide in and out of the main chamber and fit within the specs of the width of the chamber itself. Designing the bed with castors to slide it in and out of the tube was the easy part.
Th tube itself created a whole new challenge, a challenge solved with the phrase: Plastic is king.
The enclosure, a 75-centimeter diameter cylinder, is a three-layer steel chamber lined with lead, refrigerated down to near zero temperatures, and under a vacuum. Designed to nullify the earth’s magnetic field within the confines of the chamber, the introduction of metal parts inside this tube would interfere with the signal. For this reason, everything inside the chamber needed to be made of plastic, including all components of the patient bed.
Ball bearings, wheels, the scissor lift, even the adjustable support arch (the sensors needed to focus on the fetal heartbeat without mom’s heartbeat messing up the signal)—everything created for the system had to be engineered and manufactured from plastic so as not to interfere with readings.
By combining intuitive human form factor design and solid mechanical engineering solves, the delivered result by Mighty was a vision and working prototype of how this life-changing technology might exist in the future, with the hope that one day, a product such as this will be as common as MRI machine in a hospital and just another part of the pregnancy monitoring process.
When it comes to demonstrating the potential of such technology, the proof is in the prototype.